(GS3: Climate change)
The ozone ‘hole’, once considered to be the gravest danger to planetary life, is now expected to be completely repaired by 2066, a scientific assessment has suggested.
- An ozone molecule consists of three oxygen atoms instead of the usual two (the oxygen we breathe, O2, makes up 21% of the atmosphere).
- It only exists in the atmosphere in trace quantities (less than 0.001%), but its effects are very important.
- Ozone molecules are created by the interaction of ultra-violet (UV) radiation from the Sun with O2 molecules.
- Because UV radiation is more intense at higher altitudes where the air is thinner, it is in the stratosphere where most of the ozone is produced, giving rise to what is called the ‘ozone layer’.
- The ozone layer, containing over 90% of all atmospheric ozone, extends between about 10 and 40km altitude, peaking at about 25km in Stratosphere.
Need :
- The ozone layer is very important for life on Earth because it has the property of absorbing the most damaging form of UV radiation, UV-B radiation which has a wavelength of between 280 and 315 nanometres.
- As UV radiation is absorbed by ozone in the stratosphere, it heats up the surrounding air to produce the stratospheric temperature inversion.
Ozone Hole:
- Each year for the past few decades during the Southern Hemisphere spring, chemical reactions involving chlorine and bromine cause ozone in the southern polar region to be destroyed rapidly and severely.
- The Dobson Unit (DU) is the unit of measure for total ozone.
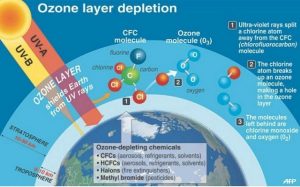
- The chemicals involved ozone depletion are chlorofluorocarbons (CFCs for short), halons, and carbon tetrachloride.
- They are used for a wide range of applications, including refrigeration, air conditioning, foam packaging, and making aerosol spray cans.
- The ozone-depleted region is known as the “ozone hole”
Tropical Ozone Hole
- According to the study, the ozone hole is located at altitudes of 10-25 km over the tropics.
- This hole is about seven times larger than Antarctica, the study suggested.
- It also appears across all seasons, unlike that of Antarctica, which is visible only in the spring.
- The hole has become significant since the 1980s. But it was not discovered until this study.
Causes:
- Studies suggested another mechanism of ozone depletion: Cosmic rays.
- Chlorofluorocarbon’s (CFC) role in depleting the ozone layer is well-documented.
- The tropical stratosphere recorded a low temperature of 190-200 Kelvin (K).
- This can explain why the tropical ozone hole is constantly formed over the seasons.
Key highlights of the report:
- The ozone layer is predicted to return to 1980 levels over Antarctica by 2066, 2045 for the Arctic, and 2040 for the remainder of the planet.
- The ozone hole has been steadily improving since 2000, thanks to the Montreal Protocol’s (1987) effective implementation.
- The Montreal Protocol mandates countries to phase out production of all main ozone depleting substances (ODSs).
- The Kigali Amendment (2016) to the Montreal Protocol aims to phase down 80-90% of the hydrofluorocarbons (HFCs) currently in use by 2050.
- HFCs do not cause much ozone layer damage, which is why they were not originally prohibited, but they are extremely powerful greenhouse gases (GHGs).
- Nearly 99% of the Montreal Protocol-banned substances have now been phased out of usage, resulting in a slow but steady rebuilding of the ozone layer.
- The success of the Montreal Protocol in closing the ozone hole is widely mentioned as a model for climate action.
- However, the parallels between the eradication of ODSs and the reduction of GHGs are limited because the usage of ODSs (though widespread) was limited to a few specific industries and their replacements were readily available.


